 *Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu #Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No LrĬlick on the element to view its propertiesĮlement (sym) # Mass density oxid #'s mp bp EN Ionization Energy rad. These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity.The Periodic Table of the Elements H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La* Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac#.  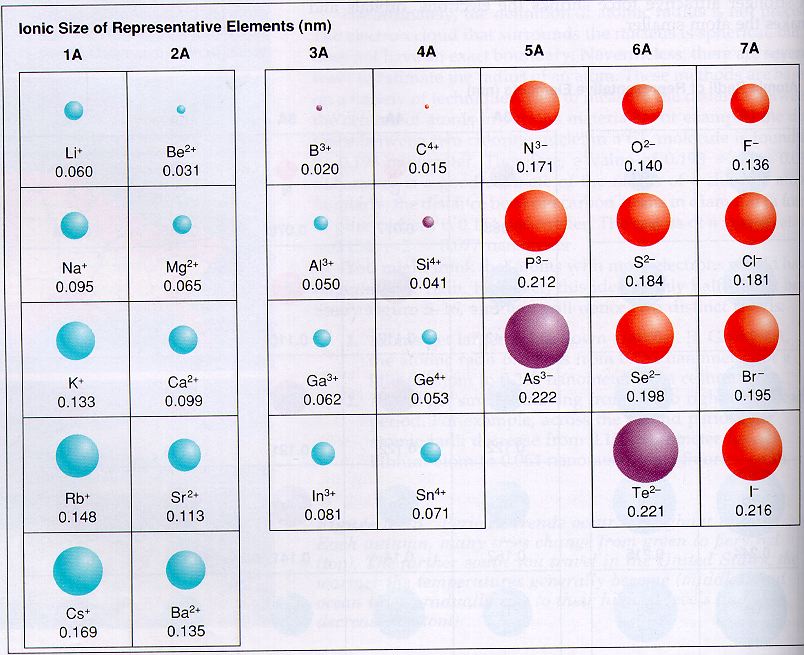
This happens because the number of filled principal energy levels (which shield the outermost electrons from attraction to the nucleus) increases downward within each group. Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. First, electrons are added one at a time moving from left to right across a period. In addition to this activity, there are two other important trends. Stable octets are seen in the inert gases, or noble gases, of Group VIII of the periodic table. 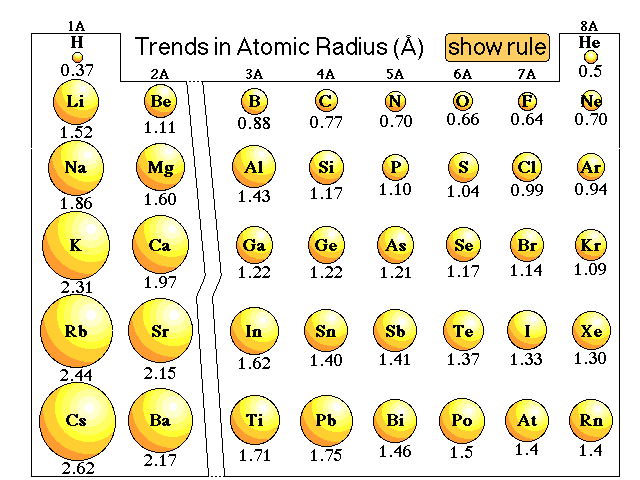 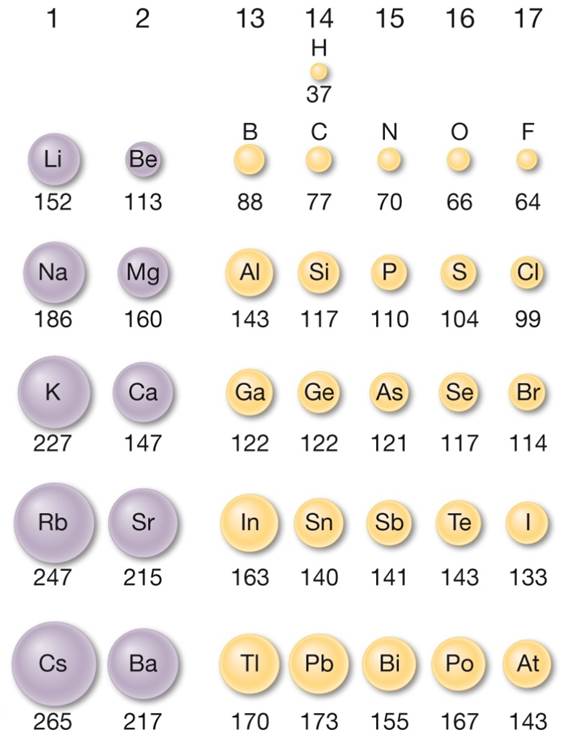
Elements tend to gain or lose valence electrons to achieve stable octet formation. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
