 By calculating the number of electron pairs around the central atom in the molecule, we can predict the molecular geometry with surprising accuracy. However, lone pairs of electrons tend to repel each other to a greater degree. By situating themselves as far as possible, it decreases the energy of the molecule, and increases the stability. This is true whether they’re bonding electrons (shared between adjacent atoms) or lone pairs (not bonded to any atom and free in space). 
The theory is underpinned by the principle of electron repulsion, which is the idea that electron pairs will naturally repel each other as far away as possible in a 3D space. VSEPR stands for Valence Shell Electron Pair Repulsion and is the most widely used model for predicting molecular geometry. We’re going to get into how VSEPR theory works and how to use it, and provide you with a simple molecular geometry chart you can easily use in order to determine your molecule’s basic shape as well as its bond angles. Although the structure of molecules can get very complicated, fortunately, there is a fairly accurate and simple way to predict the geometry of a molecule – by using the VSEPR theory. This is not only helpful in gaining a deeper understanding of the chemistry involved, but also in making assumptions about how molecules will behave under specific reaction conditions. There are a lot of ways to categorize molecules, with the aim usually being to describe and predict the chemical and physical properties accurately. 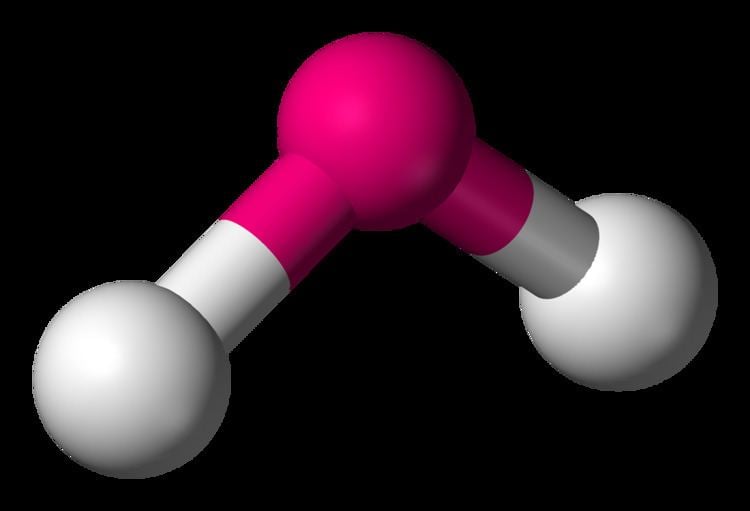
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
